Moderna COVID-19 vaccine has been granted Emergency Use Authorization EUA by the Philippine Food and Drug Administration FDA. These are as follows.

Faqs Emergency Use Authorization Department Of Health Website
9 rânduri List of authorized drugs vaccines and expanded indications for COVID-19.

Fda approved covid vaccines list. Janssen Johnson. Clinical data login required Paediatric investigation plan. 519 rânduri The FDA expanded the emergency use authorization for the Pfizer.
The three COVID-19 vaccines produced by Pfizer-BioNTech Moderna and Johnson Johnson have been granted authorization for emergency use by the FDA. Adenovirus vaccine vector ChAdOx1-S recombinant Vaccines for human use. Sinopharm SARS-CoV-2 Vaccine Vero Cell.
The FDA recognizes that vaccines are key to ending the COVID-19 pandemic and is working as quickly as possible to review applications for full approval an FDA. 104 rânduri The FDA expanded the emergency use authorization for the Pfizer-BioNTech COVID-19 Vaccine to include 12 15 year olds and issued an updated FDA COVID-19 Response At-A-Glance Summary. Two versions of AstraZeneca AZD1222.
And COVID-19 vaccine Ad26COV2S developed by Janssen Johnson Johnson on 12 March 2021. Heres the Latest Currently no coronavirus vaccine is fully approved by the FDA but three were given emergency use authorization by the agency. Spikevax previously COVID-19 Vaccine Moderna Conditional marketing authorisation granted.
F or full approval however the FDA also requires duration dat a meaning documentation of efficacy for an extended period of time. All the ones mentioned below have products that have been approved. WHO also listed the PfizerBioNTech vaccine for emergency use on 31 December 2020.
Pfizer-BioNTech COVID-19 Vaccine BNT162b2 ChAdOx1-S recombinant COVID-19 Vaccine AstraZeneca SARS-CoV-2 Vaccine Vero Cell Inactivated Coronavac Sputnik V Gam-COVID-Vac COVID-19 Vaccine. Viral Vector COVID-19 Vaccines. Vaccines for human use.
Also see the different types of COVID-19 vaccines that currently are available or are undergoing large-scale Phase 3 clinical trials in the United States. Bamlanivimab LY-CoV555 and etesevimab LY-CoV016 Immune sera and immunoglobulins for human use. Vaxzevria previously COVID-19 Vaccine AstraZeneca Conditional marketing authorisation granted.
List of COVID-19 Vaccines Authorized by the FDA. As the Delta variant surges across the country the pressure for the Food and Drug Administration FDA to grant Pfizers COVID-19 vaccine full approval is on. Healthcare providers would be able to use approved COVID-19 vaccines for off-label use.
In fact its one of just seven vaccines that the WHO has approved for inclusion on the EUL. WHO has published the target product profiles for COVID-19 vaccines which describes the preferred and minimally acceptable profiles for human vaccines for long term protection of persons at high ongoing risk of COVID-19 and for reactive use in outbreak settings with rapid onset of immunity. The Food and Drug Administration is working to approve the Pfizer - BioNTech Covid-19 vaccine on Monday The New York Times reported citing sources.
In the case of the COVID-19 vaccines currently available in the US t hat determination was based on the submission of data from carefully designed clinical trials on tens of thousands of patients. Booster shots are not yet included in Pfizer or Modernas full approval application. Are COVID Vaccines FDA Approved.
EMA recommends COVID-19 Vaccine Moderna for authorisation in the EU. Pfizer BNT162b2COMIRNATY Tozinameran INN Moderna mRNA-1273. COVID-19 mRNA Vaccines Information about mRNA vaccines generally and COVID-19 vaccines that use this new technology specifically.
Two AstraZenecaOxford COVID-19 vaccines on 15 February 2021 produced by AstraZeneca-SKBio Republic of Korea and the Serum Institute of India. There are now a total of eight 8 COVID-19 vaccines with Philippine FDA EUA approvals. Eli Lilly Canada Inc.
Authorized with terms and conditions 2021-02-26.
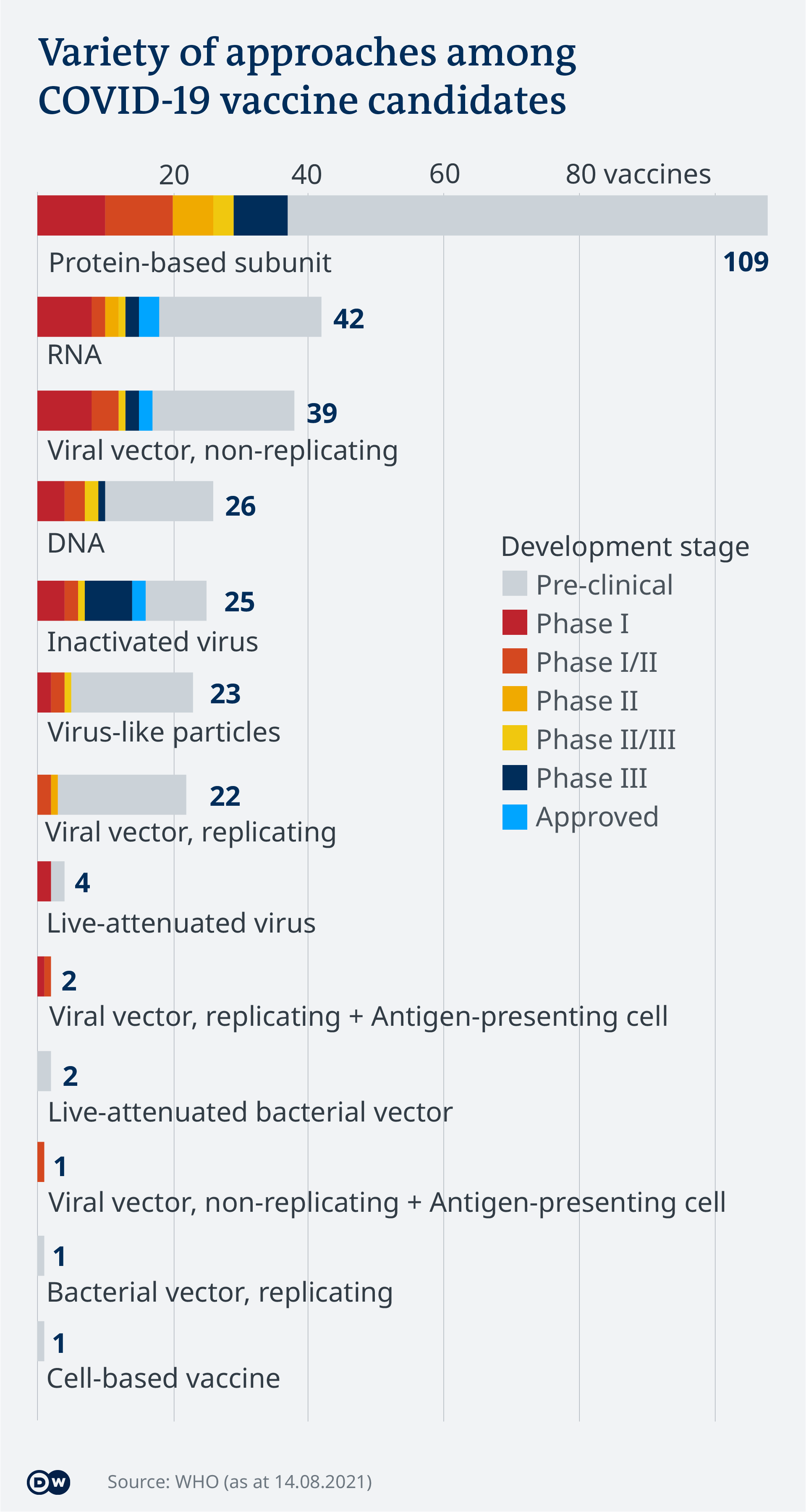
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 14 08 2021

Best Covid Vaccine To Get Comparing J J Pfizer Novavax And Moderna

Fda Advisory Panel Endorses Pfizer Biontech Covid 19 Vaccine

Novavax Announces Further Delays For Regulatory Filings Of Covid 19 Vaccine Pmlive
Covid Vaccines What Full Fda Approval Means For You

General Advisories Food And Drug Administration
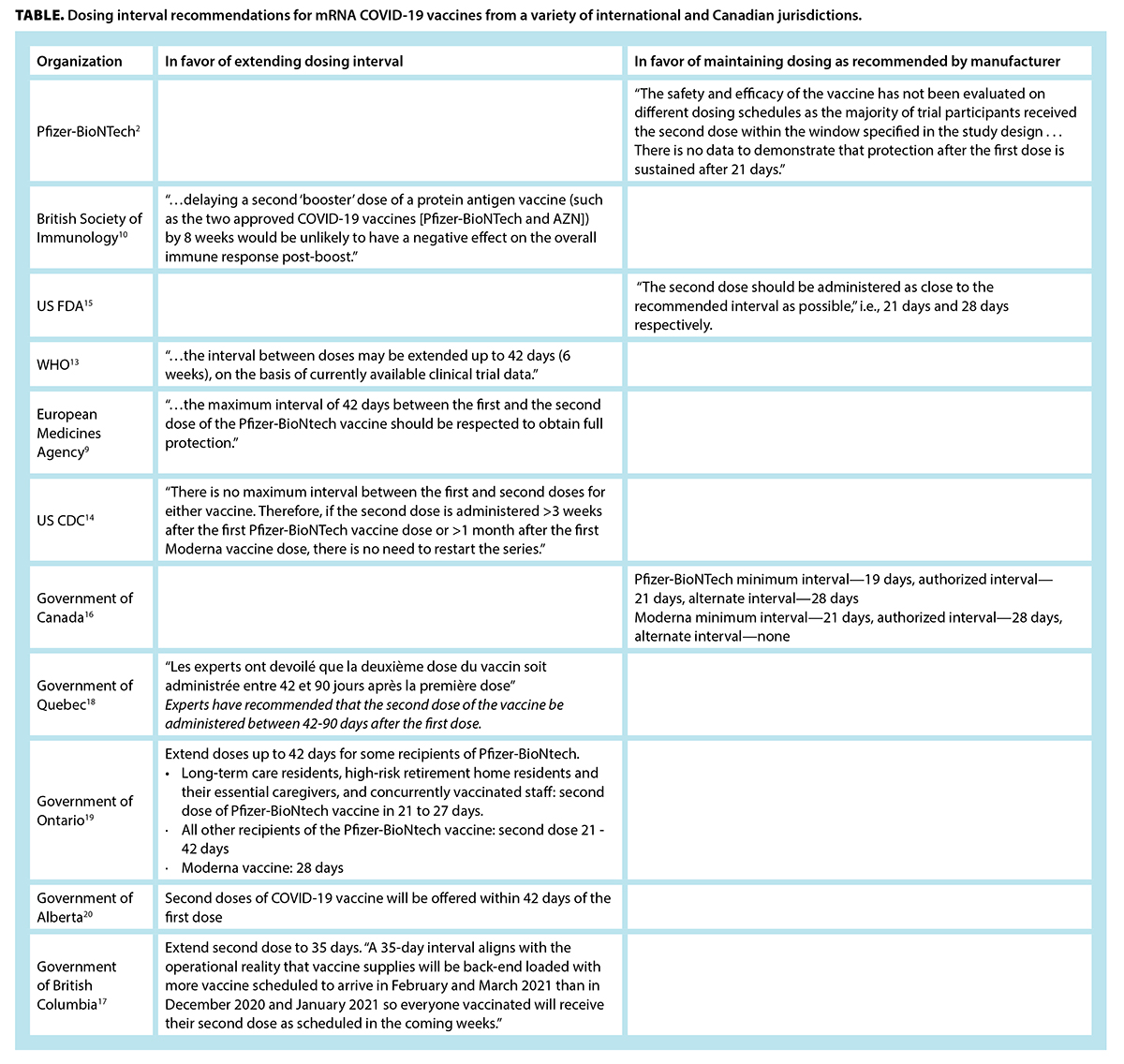
What Is The Evidence For Extending The Sars Cov 2 Covid 19 Vaccine Dosing Schedule British Columbia Medical Journal
Covid Vaccines For Kids Under 12 Expected Midwinter Fda Official Says
What Full Fda Approval Could Change About Covid Vaccination
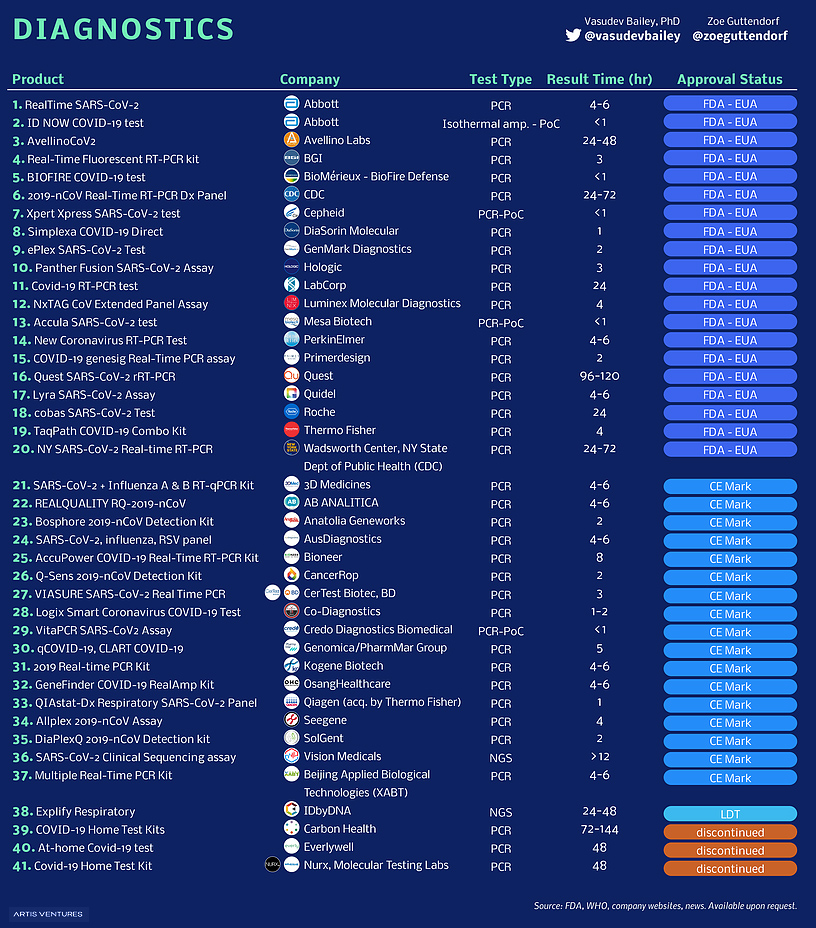
Every Vaccine And Treatment In Development For Covid 19 So Far

Know Your Vaccines Vaccine Matrix Current Evidence Department Of Health Website
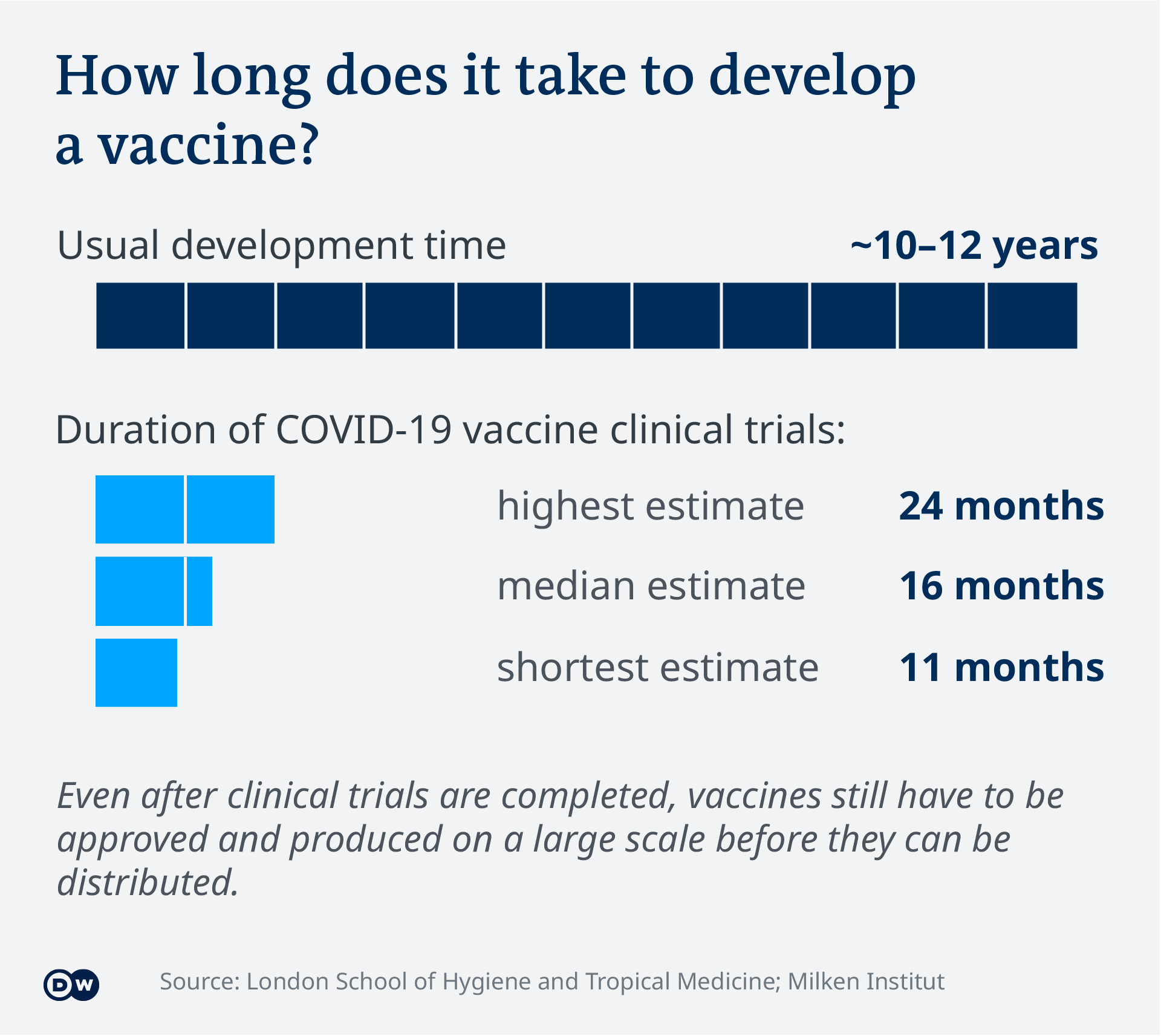
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 14 08 2021
Fda Approves J J S Single Shot Covid Vaccine For Emergency Use

Know Your Vaccines Vaccine Matrix Current Evidence Department Of Health Website

Lack Of Fda Covid Vaccine Approval Doesn T Matter Office For Science And Society Mcgill University
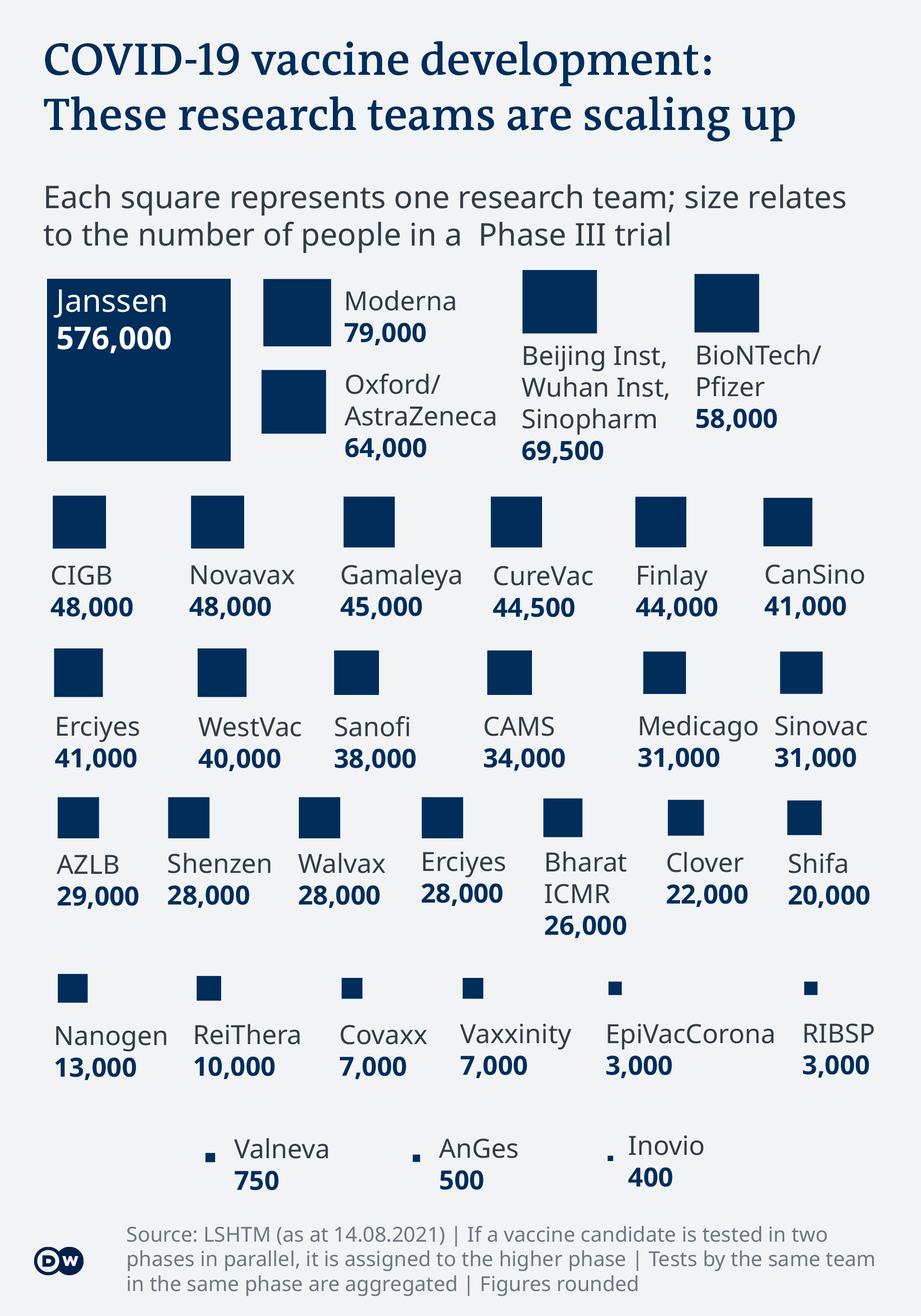
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 14 08 2021
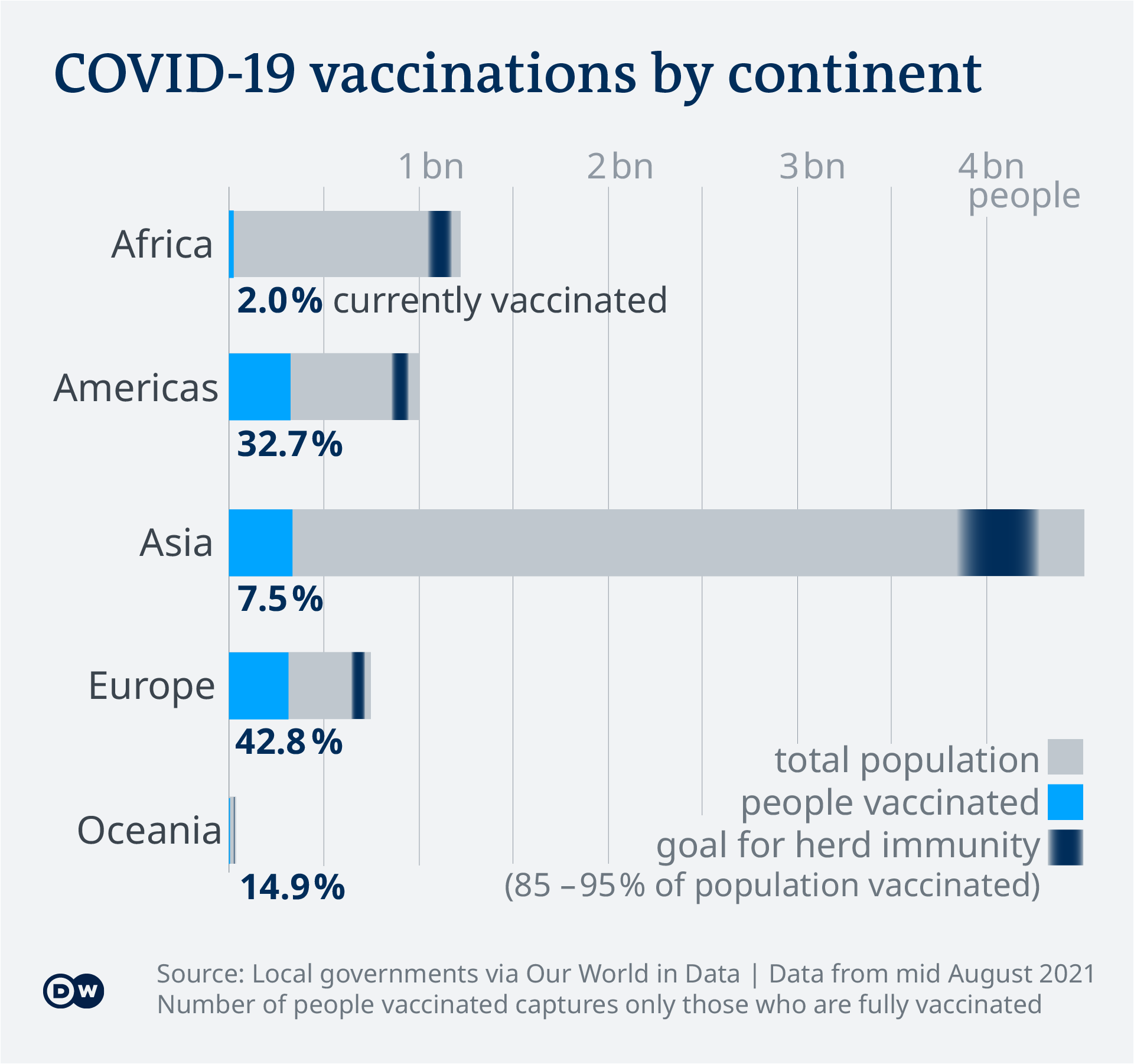
Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 14 08 2021

Fda Approval Of The Covid 19 Vaccine Could Mean More People Will Get Vaccinated For An Unexpected Reason Coronavirus Covid 19 News Fox10tv Com


